Abstract
Background: Hepatitis C virus (HCV) chronic infection has been associated with increased risk of non-Hodgkin lymphoma (NHL) in people living with human immunodeficiency virus (HIV) as well as with a trend of inferior overall survival (OS) in HIV-associated NHL in the modern antiretroviral therapy (ART) era (Besson 2020). The recent introduction of interferon (IFN)-free direct-acting antivirals (DAAs) led to the achievement of sustained virologic response (SVR) in nearly all treated patients (pts) with negligible toxicity in all settings, including HIV/HCV coinfected pts, in which, however, careful attention to interactions with ART is required. We recently showed that DAAs' administration after immuno-chemotherapy (I-CT) may improve long-term outcome in HIV-negative HCV-associated diffuse large B-cell lymphomas (DLBCL) pts (Merli 2019), however, only scant data have been reported so far about the use of DAAs in HIV/HCV coinfected NHL pts.
METHODS: We retrospectively collected clinical and virological features, treatments and outcome data of all consecutive pts with NHL and HIV/HCV co-infection, diagnosed and treated at 13 Italian centers between 2005 and 2021, with a special focus on pts affected by DLBCL and treated with DAAs. Only pts who received ART were included. The primary endpoints were SVR rate after DAAs and 2-year OS in HIV/HCV-positive NHL pts.
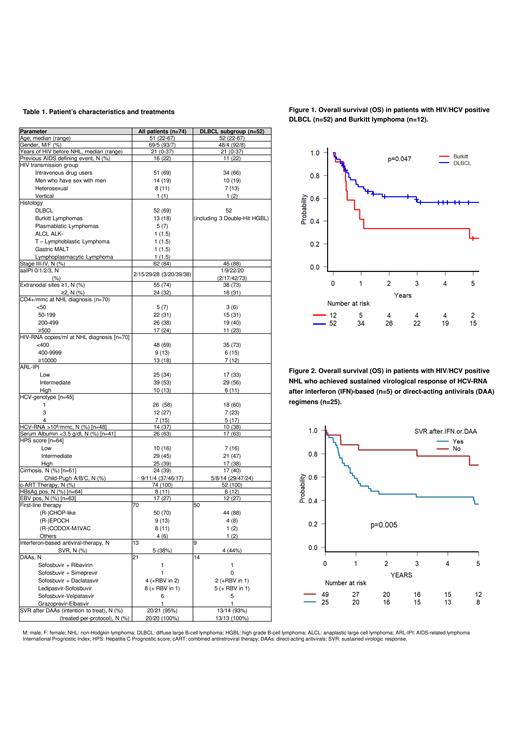
RESULTS: Overall, we collected data of 74 HIV/HCV coinfected pts with NHL (69 males, 93%), including 52 DLBCL, 13 Burkitt lymphoma (BL), 5 plasmablastic lymphoma (PL), 1 anaplastic large-cell ALK negative, 1 T lymphoblastic, 1 gastric MALT and 1 lymphoplasmacytic lymphoma (Table 1). Median age was 51 years (22-57). Previous AIDS defining event was recorded in 16 cases (22%). The main HIV transmission group was represented by intravenous drug users (69%). Stage was III-IV in 63 pts (85%) and aaIPI was ≥2 in 57 pts (77%). At NHL diagnosis 38% of pts had CD4+ <200/mmc and 31% ≥400 HIV-RNA copies/ml. ARL-IPI score was intermediate or high in 49 pts (64%). HCV genotype was 1 in 26 pts (58%), 3 in 12 (27%) and 4 in 7 (15%). Cirrhosis was present in 39% of pts (Child-Pugh B or C in 25%). Overall, 70 pts underwent curative first line therapy alongside ART, including (R-)CHOP-like in 50 (71%), (R-)EPOCH in 9 (13%), (R-)CODOX-M/IVAC in 8 (11%). Rituximab was used in 53% of cases (60% in DLBCL). 46 pts (66%) achieved a complete response (CR), 7 (10%) a partial response (PR), while 17 (24%) did not respond or progressed. At a median follow-up of 1.8 years (95%CI 0.1-12.3), 33 pts (45%) progressed, with a 2-year PFS of 53.5% (95%CI 40.7-64.8), and 38 (51%) died (30 due to NHL, 7 to infections and 1 to hepatocellular carcinoma), with a 2-year OS of 58.2% (95%CI 45.7-78.9). Two-year OS for DLBCL was 61.4% (95%CI 46.3-73.4), significantly higher than BL (39%, 95%CI 14.1-62.8; p=.0.47, Fig. 1). Considering anti-HCV therapy, 13 pts received IFN-based regimens, 5 of whom achieved SVR (38%). After 2016, 21 pts (14 DLBCL, 3 BL, 2 indolent and 2 T-cell lymphoma), including 4 who previously failed IFN, received various DAAs regimens after I-CT (sofosbuvir-based in 20). Toxicity of DAAs was minimal, with only 2 grade (G) ≥2 adverse events (1 G2 peripheral neuropathy and 1 G2 insomnia). SVR was achieved in 20/21 pts (95%): notably, the only non-responder had discontinued DAAs autonomously.
DAAs use was associated with improved OS in all pts (p=0.01) and in DLBCL (p=0.04) and with better PFS (p=0.01) in all pts. Similarly, the achievement of SVR after either DAAs or IFN predicted a better OS (Fig.2) and PFS in all pts (p=0.005 and p=0.008, respectively) and in DLBCL (p=0.018 and p=0.047, respectively). The impact of DAAs and SVR on OS remained significant also if considering only pts who achieved CR or PR after I-CT (p<0.05).
At univariate analysis, age >60 years (p=0.02), ARL-IPI (p=0.013), PS ECOG ≥2 (p=0.018) were associated with inferior OS. By applying multivariate Cox regression analysis, age >60 years (HR 67.9, 95% CI 7.2- 643.3, p<0.001), ARL-IPI (HR 2.87, 95%CI 1.03-8.06, p=0.044) and SVR after IFN or DAAs (HR 0.30, 95%CI 0.12-0.75, p=0.01) retained independent prognostic influence on OS.
CONCLUSIONS: In this very high risk series of HIV/HCV coinfected pts with NHL, mainly represented by DLBCL, the administration of DAAs after I-CT resulted feasible and effective (SVR 95%), and displayed an independent favourable influence on OS. These results strongly support DAAs' use in this hard to treat population.
Tisi: Incyte: Membership on an entity's Board of Directors or advisory committees; BWS: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees. Zilioli: Roche, Italfarmaco: Consultancy, Honoraria; MSD, Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations; Takeda: Other: travel expenses, accommodation; Gentili, Takeda, Gilead, Servier: Consultancy, Speakers Bureau. Passamonti: AbbVie: Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Arcaini: Celgene, Roche, Janssen-Cilag, Gilead: Other: Travel expenses; Bayer, Celgene, Gilead Sciences, Roche, Sandoz, Janssen-Cilag, VERASTEM: Consultancy; Gilead Sciences: Research Funding; Celgene: Speakers Bureau.